Data: 16 de junho (terça-feira) - 16:35 - 17:15
Conferências Convidadas |
| Sala Amoreira I |
|
Developing of tailored catalytic materials for the selective hydrogenation of organic molecules
Pascual Oña-Burgos
(Instituto de Tecnología Química - Universidad Politécnica de Valencia/Espanha)
|
Chair
Christian Wittee Lopes
(UFPR)
|
|
Abstract:
In recent decades, the pursuit of innovative heterogeneous catalysts composed of earth-abundant, non-precious metals has become increasingly important, with the twin goals of boosting synthetic efficiency and reducing ecological impact. Small molecule activation, such as H2O, H2, N2, O2 or CO2, constitutes a critical frontier of chemical science, potentially removing the hazardous global warming gases and providing chemical precursors from Earth's most abundant molecules.
In this talk, we shall explore how homo and heterometallic complexed can be used as building blocks to achieve different solid materials, ranging from MOFs to supported metal nanoparticles, showing enhanced efficiency in selective hydrogenation processes. The development of active and selective non-noble metal-based catalysts for the chemoselective reduction of nitrocompounds able to work in aquo media under mild conditions is an attractive research area. In fact, this reaction is key in the synthesis of derived products such as benzimidazoles, which are molecules of significant industrial interest in drug development, agriculture and material science. [1] The primary challenge in their synthesis lies in the requirement for an extremely acidic reaction medium, the employment of toxic solvents and the use of noble metals as catalysts. [2] Moreover, the single-pot formation of N-substituted pyrroles through the reductive coupling between nitrobenzene and 2,5-hexanedione is also a challenging process. Pyrroles represent a pivotal class of heterocycles found throughout bioactive natural products and man-made molecules alike, and their derivatives serve as key constituents in medicines, agrochemicals, dyes and conductive polymers. Escalating industrial demand for these structures continues to propel research toward catalytic processes that are not only efficient but also sustainable and readily scalable. [3-4] Finally, the semi-hydrogenation of alkynes to alkenes is a crucial process in the industrial purification of olefin streams. Through this process, the frequent presence of alkyne compounds as byproducts is reduced to the parts per million (ppm) range. Moreover, the production of substituted alkenes through the selective semi-hydrogenation of acetylenic compounds plays a vital role in the industrial production of polymer, pharmaceutical, and fragrance intermediates.[5] The design and development of tailored catalytic materials will face these three key hydrogenation reactions.
References
[1] J. Weber, ChemSusChem, 3 (2010), 181-187.
[2] L, Hao, Green Chemistry, 16 (2014), 3039.
[3] Y. Lu, B. A. Arndtsen. Angewandte Chemie, 47, (2008), 5430
[4] J.L. del Río-Rodríguez, S. Gutiérrez-Tarriño, L.E. Chichilla, J.P. Holgado, I.J. Villar-Garcia, V. Perez-Dieste, J.J. Calvino, P. Oña-Burgos. ChemSusChem, (2024), e202402141.
[5] I. T. Trotuş, T. Zimmermann, F. Schüth, Chem. Rev. 114, (2014), 1761–1782.
|
| Sala Amoreira II |
|
Phosphine Borane: An original ligand in organometallic chemistry and a promising organic chromophore
Ghenwa (Rinoi) Bouhadir
(Université de Toulouse)
|
Chair
Mauricio Moraes Victor
(UFBA)
|
Abstract:
We have developed phosphine–borane (PB) ligands combining donor and acceptor sites and performed extensive fundamental studies of their coordination toward transition metals (TMs).1 In particular, we have demonstrated the ability of borane to behave as a σ-acceptor ligand through unusual TM→B interactions.2 The presence of a Lewis acid in the coordination sphere of the metal drastically modifies its electronic environment and offers a unique opportunity to modulate its reactivity.3 Our recent contributions in this field will be presented, and the interplay between Lewis acids and TMs will be highlighted. 4
In addition, we recently demonstrated that phosphines are spectacular directing groups for the electrophilic C–H borylation of arenes.5 Our latest results in this area, particularly the introduction of phosphine–borane Lewis pairs onto chromophores such as pyrene and anthracene, will be presented. Remarkably, even though the phosphorus lone pair and the empty boron orbital are "neutralized" by the dative P→B bond, a significant impact on optical properties is observed.6
References:
[1] G. Bouhadir, D. Bourissou, Chem. Soc. Rev. 2016,45, 1065.
[2] For selected example, see: M. Sircoglou, S. Bontemps, G. Bouhadir, N. Saffon, K. Miqueu, W. Gu, M. Mercy, C-H. Chen, B. M. Foxman, L. Maron, O. V. Ozerov, D. Bourissou, J. Am. Chem. Soc., 2008,130, 16729.
[3] C. A. Theulier, Y. García-Rodeja, K. Miqueu, G. Bouhadir, D. Bourissou, J. Am. Chem. Soc., 2023, 145, 10800.
[4] M. Boudjelel, O. Sadek, S. Mallet-Ladeira, Y. García-Rodeja, E. D. Sosa Carrizo, K. Miqueu, G. Bouhadir, D. Bourissou, ACS Catal. 2021, 11, 3822.
[5] O. Sadek, A. Le Gac, N. Hidalgo, S. Mallet-Ladeira, K. Miqueu, G. Bouhadir, D. Bourissou, Angew. Chem. Int. Ed. 2022, 61, e202110102. doi.org/10.1002/anie.202110102.
[6] A. Le Gac, S. Mallet-Ladeira, J. Roger, J-C. Hierso, K. Miqueu, G. Bouhadir, D. Bourissou, Angew. Chem. Int. Ed. 2025, 64, e202501178. doi.org/10.1002/anie.202501178
|
| Sala Jequitiba |
|
Química de Produtos Naturais: Razão, Paixão ou Diversão?
Paulo Cezar Vieira
(FCFRP-USP)
|
Chair
Warley de Souza Borges
(UFES)
|
|
Resumo:
A investigação científica é uma atividade que envolve múltiplas competências e desafios. Nesta apresentação, discutirei alguns exemplos bem-sucedidos da minha trajetória acadêmica, desde o início na pós-graduação até a minha atuação profissional, inicialmente na Universidade Federal de São Carlos e, posteriormente, na Universidade de São Paulo.
A proposta é trazer reflexões sobre a ciência como expressão da razão, na medida em que permite compreender como as substâncias interagem, se transformam e se combinam, com base em princípios lógicos e experimentais.
Abordarei também a ciência como paixão, especialmente no contexto da química de produtos naturais, destacando o fascínio pela beleza das reações químicas e pela complexidade das moléculas, evidenciando a presença da química em nosso cotidiano — da cozinha ao desenvolvimento de novos medicamentos.
Por fim, por meio de temas instigantes e acessíveis, buscarei mostrar como é possível aprender de forma prazerosa e, ao mesmo tempo, contribuir para a construção de um mundo melhor.
|
| |
|
Data: 16 de junho (terça-feira) - 17:20 - 18:00
Conferências Convidadas |
| Sala Amoreira I |
|
Analytical Chemistry Meets AI: Opportunities and Implications
Thiago Regis Longo Cesar da Paixão
(IQUSP)
|
Chair
Paola de Azevedo Mello
(UFSM)
|
|
Abstract:
Artificial intelligence (AI) has rapidly emerged as a transformative force across the sciences, and analytical chemistry is no exception. From the automation of data treatment and spectral interpretation to the design of novel chemical sensors and intelligent microfluidic devices, AI is redefining how chemists approach analytical problems. This lecture will provide an overview of the main opportunities and challenges that AI introduces to the field, including enhanced pattern recognition, predictive modeling, and the integration of machine learning with low-cost and scalable sensing technologies. Beyond technical aspects, the presentation will also discuss the implications of AI for research integrity, education, and the future role of analytical chemists in a data-driven era. By bridging chemistry and AI, we can envision not only smarter analytical systems but also a profound redefinition of the scope and impact of analytical sciences.
|
| Sala Amoreira II |
|
Pós-COP30: Perspectivas Climáticas e o Papel da Ciência Ambiental Brasileira
Adriana Gioda
(PUC-Rio)
|
Chair
Maria Cristina Canela
(UENF)
|
|
Resumo:
Realizada em novembro de 2025, em Belém, a COP30 marcará um momento decisivo nas negociações globais sobre mudanças climáticas. Esta palestra apresentará uma análise dos principais debates e decisões da conferência, incluindo a revisão das Contribuições Nacionalmente Determinadas (NDCs), as novas metas de redução de emissões de gases de efeito estufa, estratégias de adaptação a eventos climáticos extremos, avanços no financiamento climático, e iniciativas para ampliar o uso de energias renováveis e tecnologias de baixo carbono. Serão discutidos também o papel da Amazônia, a preservação da biodiversidade e a defesa dos direitos de povos indígenas e comunidades tradicionais, temas centrais para a justiça climática. Além de apresentar os resultados e compromissos assumidos, a palestra refletirá sobre os desafios e oportunidades para a ciência ambiental e para a química brasileira na próxima década.
|
| Sala Jequitiba |
|
Synthetic Approaches for the Development of Lanthanide-Based Single-Molecule Magnets and Optical Molecular Systems
Muralee Murugesu
(Department of Chemistry and Biomolecular Sciences, University of Ottawa, Canada)
|
Chair
Fernando Sigoli
(UNICAMP)
|
Abstract: Molecules that exhibit the super-paramagnet-like property of slow magnetization relaxation and thus behave as magnets below their magnetic blocking temperature. Single-Molecule Magnets (SMMs) are promising candidates for molecular electronics such as high-density memory storage and quantum computers. Yet, the design of high-performance SMMs has been an ongoing challenge for the last three decades. This issue is particularly pronounced in multinuclear lanthanide systems, where the unpaired electrons are confined to the core 4f orbitals, limiting the possibility of strong magnetic interactions between metal centers. To address this, we have focused on incorporating small radical bridges that can enhance magnetic interactions, thereby enabling the formation of lanthanide clusters with behavior resembling the giant spin vector model typically observed in transition metal ions. Clusters based on tetrazine and pyrazine radicals exhibit strong intramolecular magnetic interactions and a large coercive fields, indicative of hard magnet behavior. These findings mark a significant advancement towards SMMs with record-high blocking temperatures. In addition, our lanthanide studies also focus on optomagnetic systems, where the interplay between optical and magnetic properties can be probed and leveraged to enhance one another.
 |
|
Data: 17 de junho (quarta-feira) - 13:00 - 13:50
Conferência Empresa - ACS |
| Sala Seringueira |
|
ACS Lunch Session: Navigating Scientific Publishing: Ask Anything to ACS Editors
|
PROGRAMA
13:00-13:05: Opening Remarks
Regiane Bracchi, MS
Regional Manager, American Chemical Society
13:05-13:40: Q&A Session with ACS Editors
Speakers TBD
13:40-13:45: Closing Remarks
Regiane Bracchi, MS
Regional Manager, American Chemical Society
13:45-14:00: Networking Lunch
|
| |
|
Data: 17 de junho (quarta-feira) - 16:35 - 17:15
Conferências Convidadas |
| Salas Jequitiba |
|
Chemical Warfare: Our Chemistry, Our History
Imee Su Martinez
(University of the Philippines - Filipinas)
|
Chair
Jéssica Fonsaca
(PUC-Rio )
|
|
Abstract:
Weaponizing chemicals is very much entwined with human history. Chemical weapons have been used for generations and is very much ingrained in people's minds. The accessibility of chemicals being a part of people's day-to-day existence makes them even more prone to misuse. The dual nature of chemicals allows them to be readily converted from friend to foe, simply by breaking and creating bonds. The ethical compass of chemical practitioners is therefore essential in ensuring that the duality of chemistry is respected, and the peaceful use of chemistry is upheld. The Chemical Weapons Convention is the most successful disarmament treaty to date, ensuring the full destruction of verified chemical weapon stockpiles. Is this the end to chemical weapons?
In this presentation, the dual nature of chemistry will be discussed through the lens of chemical warfare in human history. Moreover, the pathway to prevent the resurgence of chemical weapons will be traversed, through the careful surveillance of related science and technology, which are the underpinnings of the convention, and most importantly, through awareness and education ꟷinstilling a chemical conscience in every chemist.
|
| Sala Carvalhos I |
|
Produção Científica e Soberania Nacional: Qual a relação?
Agustina Rosa Echeverría
(UFG)
|
Chair
Bruno Silva Leite
(UFRPE)
|
|
Resumo:
A produção científica não é apenas um indicador acadêmico ou institucional, mas desempenha um papel central na constituição e manutenção da soberania nacional. Soberania envolve autonomia política, econômica e cultural, e depende fortemente da capacidade de um país gerar, distribuir e aplicar conhecimento científico de modo reflexivo e crítico. Nesta palestra, busca-se explorar como a pesquisa acadêmica contribui para essa autonomia, quais obstáculos se colocam no caminho e de que modos políticas de educação em ciências podem fortalecer esse laço.
|
| |
|
Data: 17 de junho (quarta-feira) - 17:20 - 18:00
Conferências Convidadas |
| Sala Amoreira I |
|
Oxonium Ions, Total Synthesis and Structure Determination
Jonathan W. Burton
(University of Oxford)
|
Chair
Emilio Carlos de Lucca Júnior
(UNICAMP)
|
Abstract:
The primary focus of this presentation will be the chemistry of oxonium ions; additionally, the total synthesis of numerous natural products along with the structure reassignment of a number of natural products will be presented.
Trialkyl oxonium ions have been proposed as intermediates in the biosynthesis of numerous C15-halogenated natural products from Laurencia species.1,2 Previously we have reported the synthesis and characterization (low T NMR) of four of these trialkyl oxonium ions, and demonstrated their reaction with a range of nucleophiles, at two of the three electrophilic sites (C-10 and C-7), to yield numerous natural products; notably the reaction of nucleophiles at C-13 was not observed (Figure 1).3,4
In this presentation we report the synthesis, characterization (low T NMR) and reactivity of a proposed biosynthetic allyldialkyl oxonium ion. This oxonium ion reacts with nucleophiles primarily at the allylic position (C-13) leading to a plausible biosynthetic route to four dioxabicyclo[2.2.1]heptane Laurencia natural products.
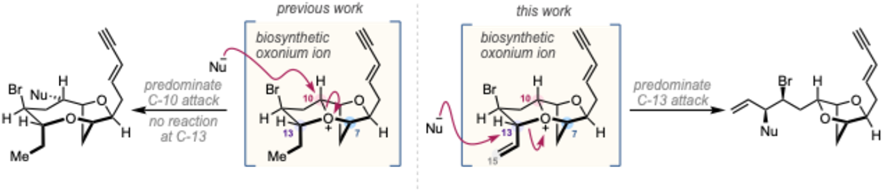 Figure 1. Figure 1. Site selectivity in the reaction of related oxonium ions with nucleophiles.
References
[1] A. Fukuzawa, M. Aye, M. Nakamura, M. Tamura, A. Murai, Tetrahedron Lett. 1990, 31, 4895.
[2] H. Kikuchi, T. Suzuki, E. Kurosawa, M. Suzuki, Bull. Chem. Soc. Jpn. 1991, 64, 1763.
[3] H. S. S. Chan, Q. N. N. Nguyen, R. S. Paton, J. W. Burton, J. Am. Chem. Soc. 2019, 141, 15951.
[4] H. S. S. Chan, A. L. Thompson, K. E. Christensen, J. W. Burton, Chem. Sci. 2020, 11, 11592.
|
| Sala Amoreira II |
|
Descoberta de Fármacos inspirada pela natureza: Estratégias multidisciplinares em química e biologia para a identificação de novos compostos líderes para a malária
Rafael Victorio Carvalho Guido
(IFSC-USP)
|
Chair
Floriano P. da Silva Jr.
(FIOCRUZ)
|
|
Resumo:
A malária é uma parasitose de distribuição global com elevada mortalidade, apesar dos avanços e investimentos em novas terapias. Atualmente, a doença permanece endêmica em aproximadamente 80 países, tendo registrado cerca de 282 milhões de casos e 610 mil óbitos em 2024. Nesse contexto, produtos naturais provenientes da biodiversidade brasileira, bem como seus derivados sintéticos, destacam-se como fontes estratégicas para a identificação de potenciais candidatos a fármacos antimaláricos. Esta conferência abordará as estratégias integradas em química de produtos naturais, síntese orgânica, química medicinal, biologia celular e biologia estrutural empregadas pelo grupo de pesquisa na descoberta de novos compostos líderes para o tratamento da malária.
Title: Nature-driven Drug Discovery: Multidisciplinary Chemistry and Biology Approaches for Identifying New Lead Candidates for Malaria
Abstract:
Malaria is a globally distributed parasitic disease with a high mortality rate, despite significant advances and investments in new therapies. The disease is endemic in approximately 80 countries, with around 282 million cases and 610,000 deaths reported in 2024. In this context, natural products sourced from Brazilian biodiversity, as well as their synthetic derivatives, are considered strategic resources for identifying potential antimalarial drug candidates. This conference will explore the integrated strategies in natural products chemistry, organic synthesis, medicinal chemistry, cellular biology, and structural biology employed by the research group to discover new lead compounds for malaria treatment.
|
| Sala Jequitiba |
|
Ciência ou Crença?
Como a informação correta transforma realidades
Gabriela Bailas
(Divulgadora científica @bibibailas)
|
|
Em breve mais informações
|
| Sala Carvalhos I |
|
Estratégias computacionais para o desenvolvimento de fotossensibilizadores: da mecânica quântica às técnicas de machine learning
Paula Homem-de-Mello
(CCNH-UFABC)
|
Chair
Hueder Paulo Moisés de Oliveira
(UFABC)
|
|
Resumo:
Fotossensibilizadores têm diversas aplicações, desde sensores e células solares a tratamentos médicos. É importante, portanto, propor novas moléculas que absorvam radiação na região do visível do espectro eletromagnético e que possam ser ajustadas para interagir com os diferentes alvos de maneira eficiente. Algumas das estratégias envolvem o estudo dos processos fotofísicos e fotoquímicos em detalhe, de forma a descrever com acuracidade a energética e o mecanismo. Por outro lado, também é possível trabalhar com conjuntos de milhões de moléculas já conhecidas e obter descritores que ajudam no planejamento de novos compostos. Assim, nesta conferência, serão apresentadas diversas abordagens computacionais, desde métodos baseados na mecânica quântica a técnicas de machine learning, que têm sido empregadas no design de novas moléculas e materiais.
|
|

